 Molecules with mirror symmetry like oxygen, nitrogen, carbon dioxide, and carbon tetrachloride have no permanent dipole moments. As you can see, ammonia consists of one nitrogen atom covalently bonded to three hydrogen atoms. For example, ammonia (NHsub3) is a polar molecule. Their vector sum is zero, so CO2 therefore has no net dipole.Ī dipole moment is simply the measure of net polarity in a molecule. Because the two C–O bond dipoles in CO 2 are equal in magnitude and oriented at 180° to each other, they cancel. H2O has dipole moments, but they do not cancel out because they are equal but not opposite in direction.Įach C–O bond in CO 2 is polar, yet experiments show that the CO 2 molecule has no dipole moment. Therefore, CO2 is a nonpolar molecule despite having polar bonds. What is dipole moment out of water and carbon dioxide which is polar and why?ĬO2 has dipole moments, but they cancel out because they are equal in magnitude and opposite in direction. In H2O, Oxygen is more electronegative, so the dipole moments would face inwards towards oxygen and consequently cancel out. Keep in mind that in CO2, Oxygen is more electronegative, so the dipole moments are facing opposite directions (away from the central atom of Carbon), making the molecule polar. In the case of water, oxygen is more electronegative than hydrogen, so both dipoles point from hydrogen to oxygen. According to an electronegativity chart, oxygen is more electronegative than carbon, so we draw our dipoles pointing from carbon to oxygen. In the case of carbon dioxide, we have two dipoles. Why does the water molecule have a dipole and why doesn’t the carbon dioxide molecule have one? 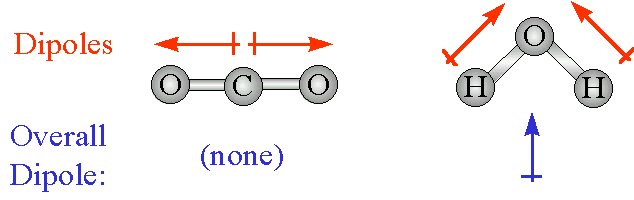
It is because water has been a bent structure in which two O-H bonds are oriented at an angle of 104.5° and do not cancel the dipole moments of each other. The CO2 molecule has a zero dipole moment even though C and O have different electronegativities and each of the C = O bond is polar and has the same dipole moment. 
Why is the dipole moment of CO2 is zero while H2O is a polar though both have similar formula? 5 Why does CO2 have no dipole moment but SO2 has?.2 What is dipole moment out of water and carbon dioxide which is polar and why?.1 Why is the dipole moment of CO2 is zero while H2O is a polar though both have similar formula?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
